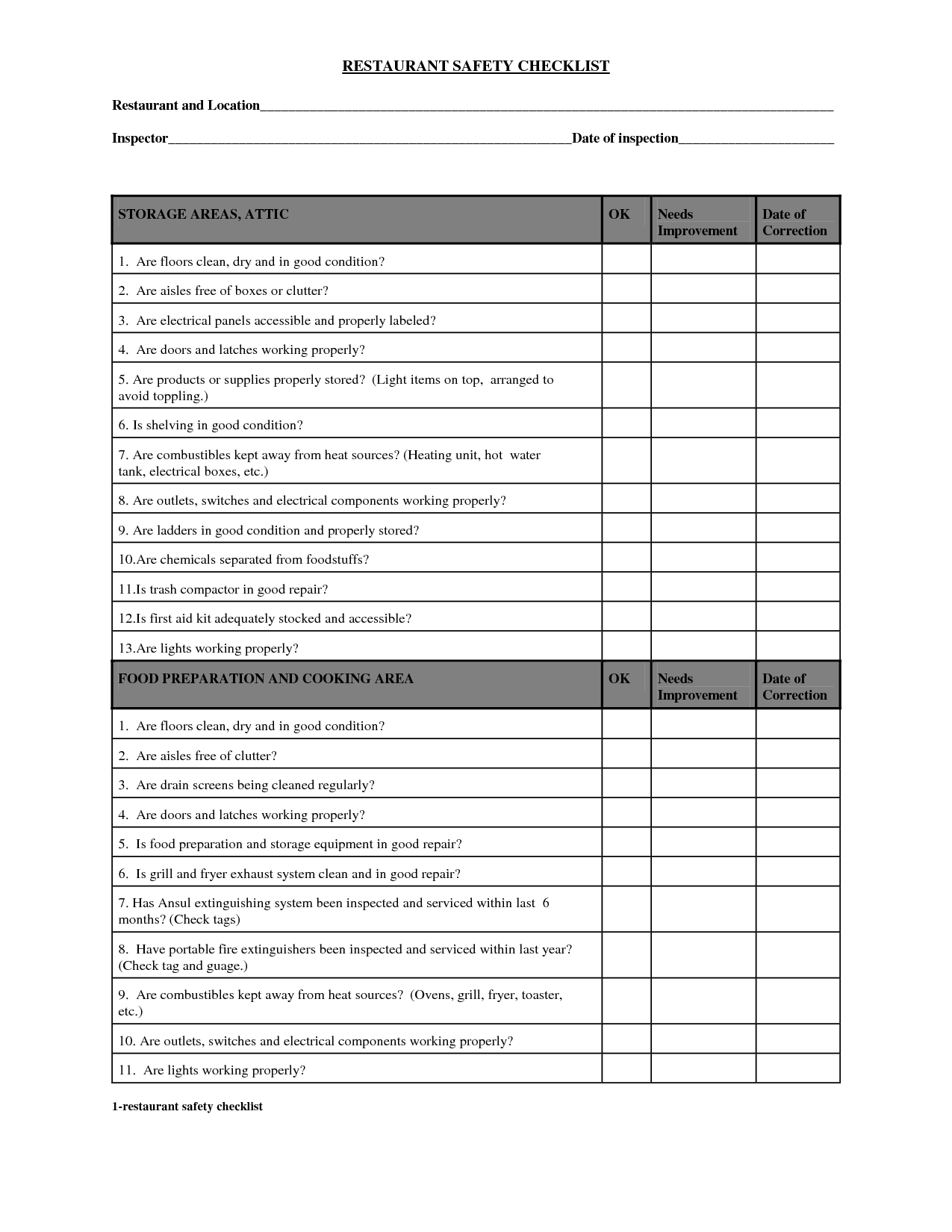
Nsf Audit Checklist3/22/2021
Subscribe to the blog at furfarmandfork.com for food safety research, insights, and analysis.
Nsf Audit Checklist Code That HasI would like to ask, if we were previously SQF Code Edition 7.2 (Level 3, July 2014) certified, which system elements is applicable for us I am confused because the new SQF Edition 8 has Quality Code and another is in Manufacturing code that has the system element.Can I say that we will be using the checklist for quality and also for the manufacturing system element Awaiting for any inputs.
We connect those interested in food safety to information and to each other. The purpose of the website is to share knowledge, experiences and ideas to make food safer. If you continue without changing your browser settings, well assume that you are happy to receive all cookies on our website. Performing regular GMP audits protects a business from product safety issues, product recalls, and legal and regulatory problems. Auditing Tool for GMP Compliance Conduct a thorough and systematic GMP audit using a digital checklist. Use iAuditor, the worlds 1 inspection app, to replace existing workflows involving paper forms, spreadsheets, scanning and faxing, and perform audits on your mobile phone or tablet. With iAuditor you can: Create mobile-ready GMP audit checklists Empower teams to complete GMP audits using a smartphone or tablet Attach photos and make annotations within the app Save all reports online. Use for free with small teams. Unlimited reports and storage for premium accounts Compare pricing plans. Best GMP Compliance Checklists GMP Compliance Checklist A GMP Compliance Checklist is used to evaluate a manufacturing companys compliance with manufacturing protocols. Use this checklist to perform a facility walkthrough and manufacturing observation of all 8 relevant systems: 1) Building and Facilities; 2) Materials Management; 3) Quality Control Systems; 4) Manufacturing; 5) Packaging and Identification Labeling; 6) Quality Management Systems; 7) Personnel and Training; and 8) Purchasing and Customer Service. Use iAuditor for your GMP audits to: Perform digital inspectionsaudits with your mobile phone or tablet Capture photo evidence of compliant and non-compliant items Add corrective actions for immediate resolution on identified non-compliant items Generate comprehensive reports as you finish an audit. Preview sample report here Save reports online and easily share it with members of your organization Promote accountability for inspections done by capturing digital signatures Download Template Preview Report GMP Food Manufacturing Audit Checklist This GMP Food Manufacturing Audit is based on the 21 CFR Part 110 checklist and can be used as an assessment tool to ensure your organization follows manufacturing standards. Identify problem areas and assign immediate corrective actions by performing your food manufacturing audits with this template using the iAuditor mobile app. Securely save your completed reports in the cloud and track overall audit scores and performance. Nsf Audit Checklist Full Facility WalkthroughDownload Template GMP Site Walkthrough Checklist A GMP Site Walkthrough Checklist is a tool used in conducting a full facility walkthrough and observation. Use this checklist to check a manufacturing sites overall safety by assessing pest control, operational methods practices, PPE, building ground areas, and more. Identify and take photos of potential hazards and assign corrective actions to authorized personnel for immediate resolution. Download Template FDA GMP Cosmetics Audit Checklist This digital GMP Cosmetics Checklist has been built using the iAuditor app and based on the FDAs GMP Cosmetic Guidelines. Use this digital GMP checklist to assess your manufacturing compliance with FDA guidelines across aspects of building and facilities, equipment, personnel, raw materials, production, lab controls, record keeping, labelling and complaints. Use iAuditor to take notes and photo evidence of non-compliance. Automatically calculate your audit score based on the responses and generate a complete report without leaving your manufacturing facility. Download Template Good Distribution Practice (GDP) Self-Inspection Checklist Use this GDP self inspection checklist to assess compliance with EU Good Distribution Practice guidelines. This template covers questions across the areas of quality systems, personnel, premises and equipment, documentation, supplier operations, complaints recalls and transportation. Calculate overall GDP compliance score across sections, identify frequently failing items and take photos and notes of non-compliance. Nsf Audit Checklist Trial Safety ChecklistDownload Template Related pages: Checklist Waterproofing Inspection Industrial Safety Checklist Brewery Safety Manufacturing Facility Reopening Manufacturing Process Audit Commissioning Checklists ITAR Compliance Process Validation Report OEE Report More Guide GMP Office Safety and Health Comprehensive Guide Office Safety OSHA Standards Food Safety More App Manufacturing Inspection Software Layered Process Audit Software First Article Inspection App Forklift App Field Inspection App More Start your free account today Getting started is easy, simply fill in your email and raise the game with iAuditor Your work email Get started with iAuditor today Were sorry, something went wrong with the submission. Back to top Product Product Inspection Software Temperature logging TeamTrain Spotlight Docs by Safetyculture Checklist App Checklist Maker Resources Resources Templates Checklists Topics Blog Events Webinars Apps API Support Support Support center Product updates Book a demo Integrations Company Company About us Careers Meet the team Contact us Product Inspection Software Temperature logging TeamTrain Spotlight Docs by Safetyculture Checklist App Checklist Maker Resources Templates Checklists Topics Blog Events Webinars Apps API Support Support center Product updates Book a demo Integrations Company About us Careers Meet the team Contact us Status Privacy Terms conditions Security English SafetyCulture 2020.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
